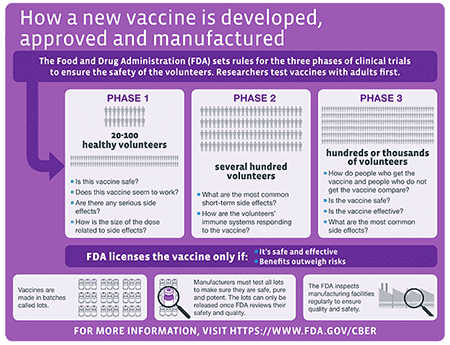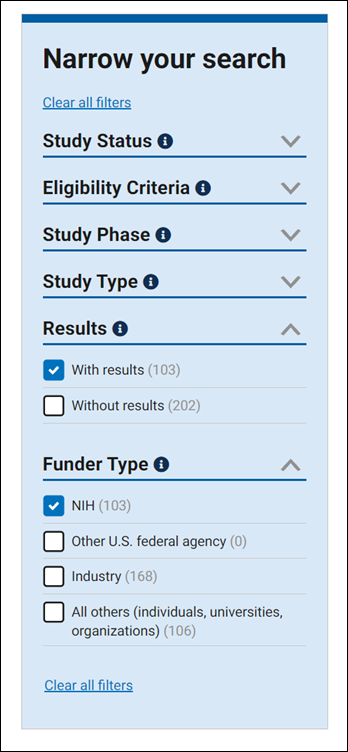
Beta ClinicalTrials.gov Website Released - Let Us Know What You Think!. NLM Technical Bulletin. 2021 Nov–Dec

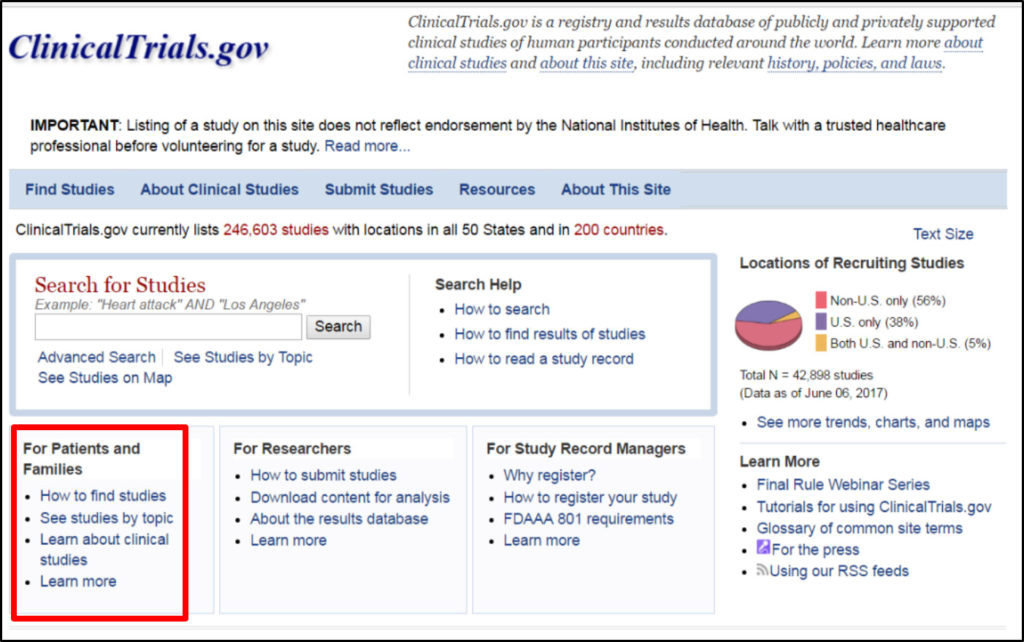
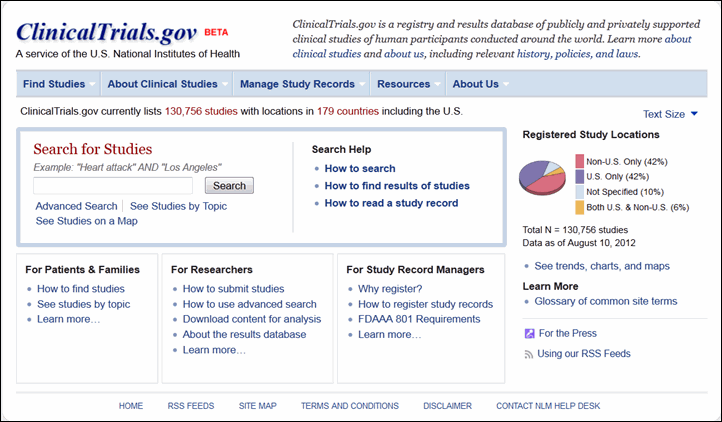
Celebrating 20 Years of ClinicalTrials.gov and Looking to the Future – NLM Musings from the Mezzanine

Compliance with legal requirement to report clinical trial results on ClinicalTrials.gov: a cohort study - The Lancet

COVID Trial Finder Provides Simplified Search Process For COVID-Related Clinical Trials - Columbia DBMI
How Frequently Do the Results from Completed US Clinical Trials Enter the Public Domain? - A Statistical Analysis of the ClinicalTrials.gov Database | PLOS ONE

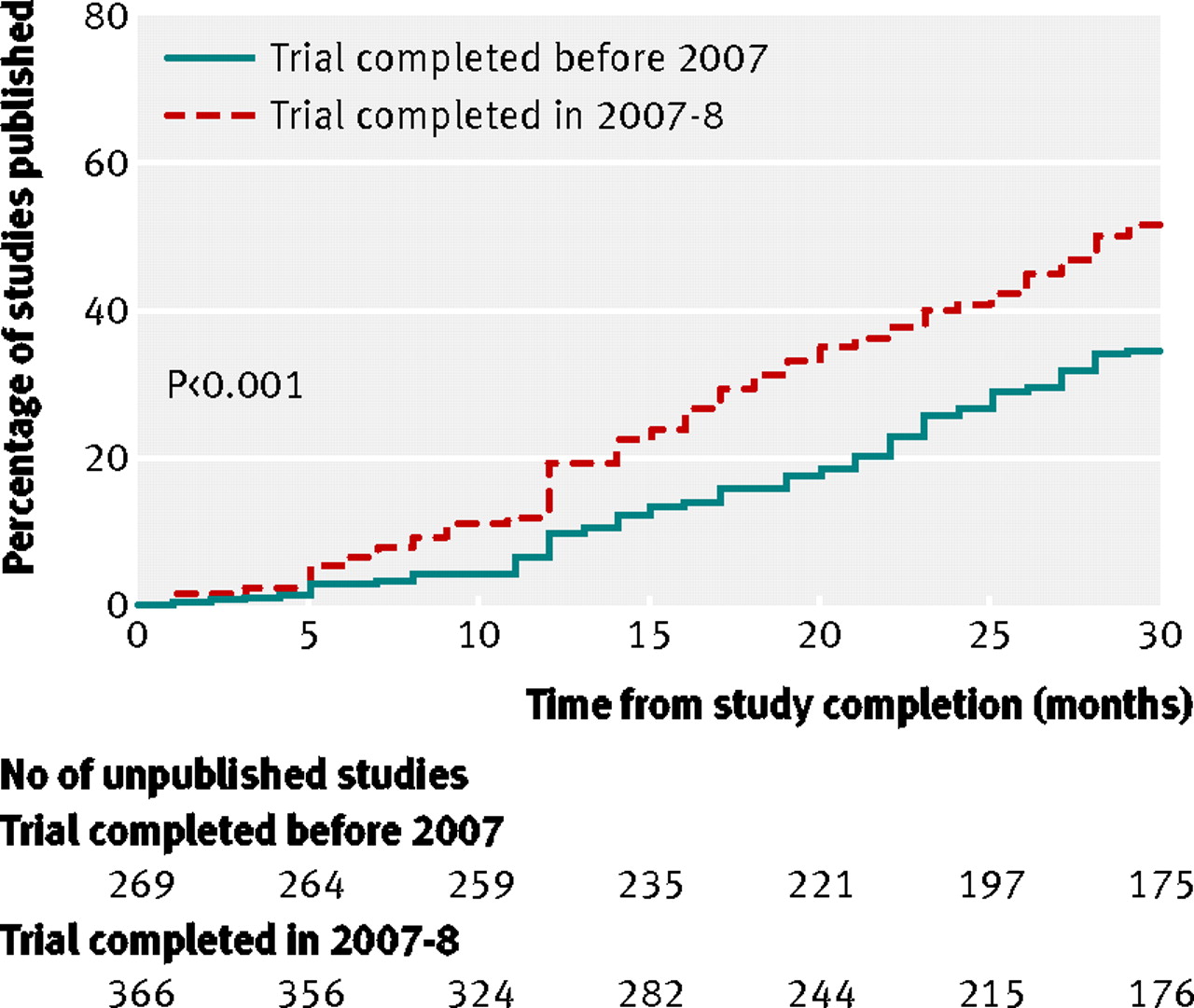
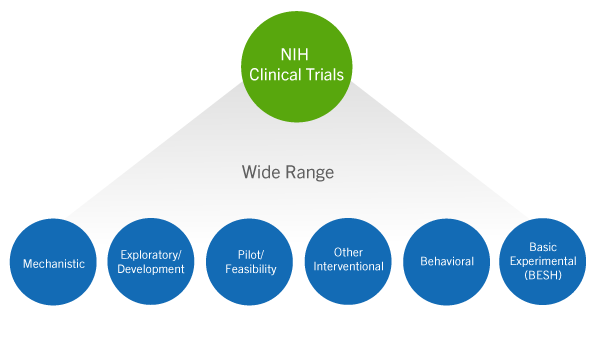
Publication of NIH funded trials registered in ClinicalTrials.gov: cross sectional analysis | The BMJ

Publication of NIH funded trials registered in ClinicalTrials.gov: cross sectional analysis | The BMJ