Metformina teva italia 1000 mg compresse rivestite con film - 1.000 mg compresse rivestite con film 60 compresse

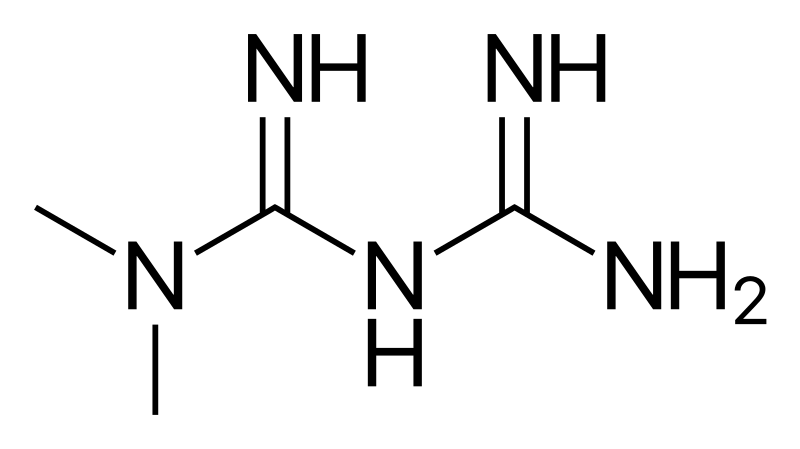
Teva Pharmaceuticals USA, Inc. Initiates Voluntary Nationwide Recall of Metformin Hydrochloride Extended-Release Tablets USP 500 mg and 750 mg Due to Detection of N-Nitrosodimethylamine (NDMA) | FDA