Amazon.com: The Four Villains of Clinical Trial Agreement Delays and How to Defeat Them: Addressing CTA Delays Comprehensively: 9781973622697: Araujo, Débora S.: Books
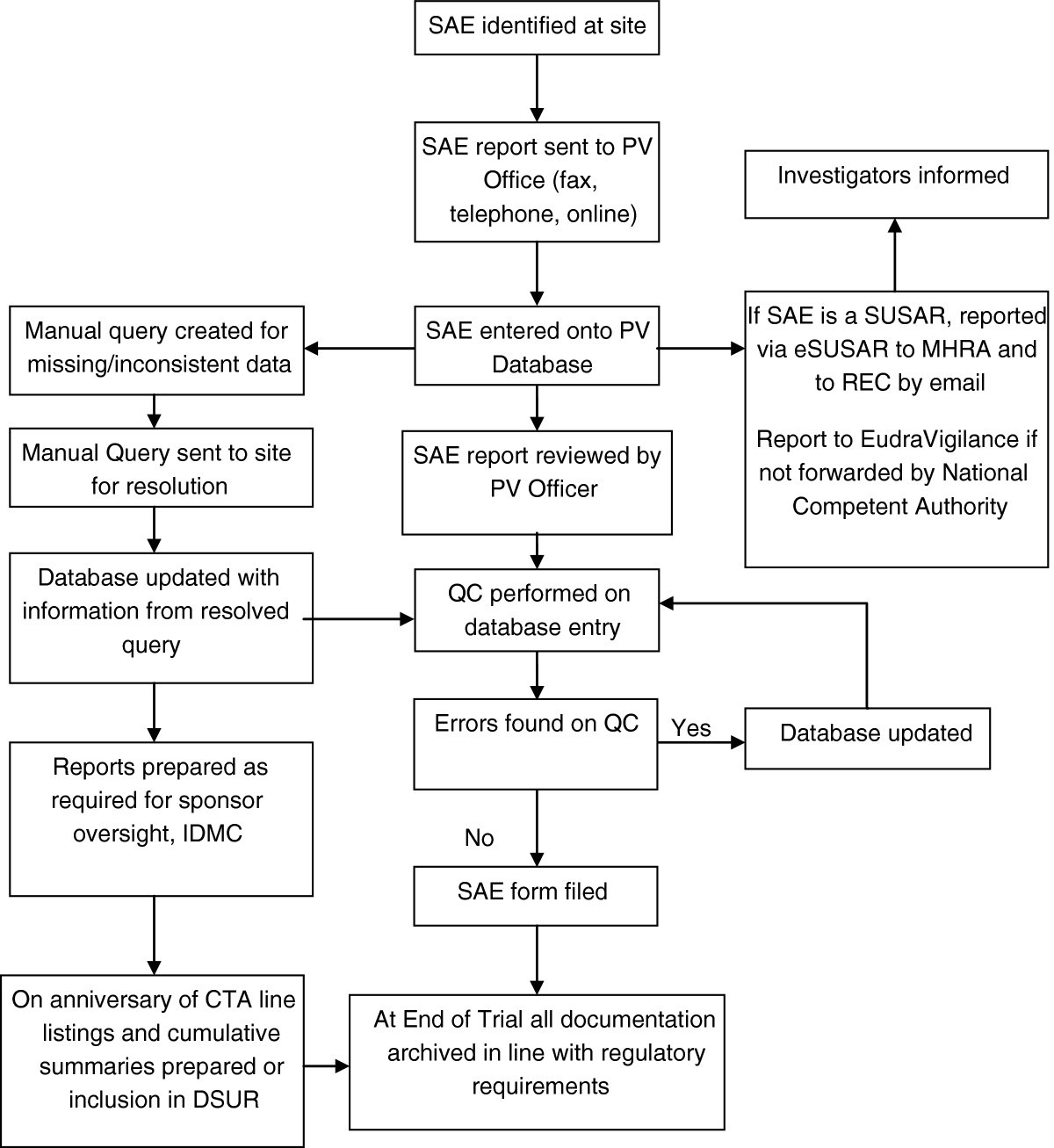
Implementing a centralised pharmacovigilance service in a non-commercial setting in the United Kingdom | Trials | Full Text

QurAlis Receives Clinical Trial Authorisation (CTA) in European Union (EU) for QRL-201, a First-in-Class STATHMIN-2 Precision Therapy for ALS